http://www.hyle.org
Copyright © 2014 by HYLE and Mi Gyung Kim
Stabilizing Chemical Reality: The Analytic-Synthetic Ideal of Chemical SpeciesMi Gyung Kim*
1. IntroductionThere has been a long debate over whether chemistry is an art or a science and, as a corollary, whether chemical products are artificial or natural (Bensaude-Vincent & Newman 2007, Bensaude-Vincent & Simon 2008). Now that we have synthesized over twenty million chemical species and are making a brave new world through synthetic biology, it might be useful to rehash this philosophical quagmire. Making new worlds, as chemists have been doing for centuries, often depends on decomposition and composition (Goodman 1978, pp. 7-10). Synthetic biologists thus insist that their field was conceived as "a program to produce a set of well-characterized and systematized biological components that can be generically assembled to create custom biological circuitry" (Gardner & Hawkins 2013, added emphasis). If the "standardization of reusable biological components is the most efficient and effective way to engineer" biological species (ibid.), it may not be an accident that this opinion appeared in a journal bearing the title of chemical biology.[1] These statements could just as easily summarize chemists’ stabilization of elements and compounds in the eighteenth century and their subsequent pursuit of organic synthesis in the nineteenth century. As Thomas S. Kuhn observed, chemical atomism required "a belief in the endurance of elements in their compounds" and the recognition of "analysis and synthesis as fundamental tools of the working chemist" (Kuhn 1952, p. 13, added emphasis). While perceptively identifying the foundational tenet of modern chemistry as the relationship between elements and compounds, he did not quite appreciate the material contingency of chemical substances that depended on chemist’s historically specific tools and methods. Chemistry and synthetic biology make new laboratory objects and hybridize them as an accepted part of the historically contingent material world, which integrates them as a part of the ‘true’ (but historically contingent) representation of ‘nature’. Chemists acquired the power to make and re-make their worlds much earlier than other scientific practitioners and can serve as a model discipline in examining scientific ‘world-making’ in Nelson Goodman’s term. Their remarkable success in stabilizing and universalizing their laboratory reality depended critically on their dogged pursuit and promotion of the analytic-synthetic ideal of chemical substances.[2] This was a practical commitment to maintain their disciplinary practice and identity, rather than a philosophical idea, although they often naturalized it through dominant philosophical languages at least until the 1830s. This paper points to the central importance of analytic (or synthetic) methods in producing, stabilizing, and universalizing the objects and theories of chemistry, which has maintained its disciplinary identity and terrain. A sudden advancement in chemical practice is more often caused by a radical innovation in analytic method than by a new theory.[3] Effective chemical theories such as the affinity table and structural formula took stock of the analytic advances to construct an intelligible representation of the chemical world for a desired audience. If this dialectic between analytic methods and (often classificatory) theories characterizes chemistry’s evolutionary path from the late seventeenth century, which has produced and stabilized pure chemical substances, the need for new theoretical structures became acute at certain moments for technical and social reasons. During these contentious moments, chemists had to scrutinize their taken-for-granted analytic methods to construct a more coherent representation of chemical reality through a new theoretical structure that could coordinate their practice and persuade a broader audience. While early modern chemists sought to reconcile their laboratory reality with the dominant philosophical discourse of nature to authenticate its realness for the literate public, modern chemists could make them real by proving their industrial, commercial, or military utility. In forcing their laboratory reality to subsist in the outside world, they not only stabilized chemical species as ‘natural kinds’ but also hybridized the ‘nature’ that came under scientific scrutiny. By focusing on these theoretical moments when the productivity of analytic methods challenged the existing theory’s function, purpose, and efficacy, we can appreciate the centrality of analytic methods in mapping and re-mapping chemistry’s disciplinary terrain and laboratory reality.[4] To this end, this paper will configure a series of theoretical moments in the development of French chemistry to underscore how difficult it was for chemists to coordinate their constantly mutating analytic methods, to produce a reliable laboratory reality, and to package it as a true representation of nature. As I have outlined in Affinity, That Elusive Dream (Kim 2003), the first theoretical moment developed at the turn of the eighteenth century when a cluster of Paris Academicians sought to craft a coherent chemical reality and representation by coordinating distillation and solution methods. Although some sought to naturalize chemical theory in the newly dominant Cartesian philosophy, others charted experimental pathways to produce the affinity table that organized the ‘chemistry of salts’ (Holmes 1989a) as something like Kuhn’s ‘normal science’ and stabilized the analytic-synthetic ideal of chemical species. During the second theoretical moment, conventionally known as the Chemical Revolution, Lavoisier introduced metric measurements and algebraic representation to refine the analytic-synthetic ideal while incorporating the gases. Resistance to his reform makes more sense at this instrumental-epistemological level than at the level of ontology (Kim 2008, 2011, Chang 2012). His justification of the new nomenclature in Condillac’s philosophy changed chemistry’s philosophical association from metaphysics to epistemology, an emergent philosophical concern that would fundamentally refocus academic philosophy (Gadamer 1993, p. 220; Rorty 1979, pp. 131-164; Kim 2009).[5] The early nineteenth-century development of stoichiometry, which led to a sharper analytic resolution of organic compounds, prepared for the third theoretical moment in the 1830s when chemists struggled to invent a rational classification of organic substances (Kim 1992a/b, 1996). By this time, they could dispense with philosophical justification – naturalizing chemical substances through metaphysics – and lay claim to truthful representation and direct transformation of nature through laboratory methods, which indicates the changing fortunes of philosophy and chemistry (Dumas 1837, Bensaude-Vincent 1999). Only with the stabilization of pure chemical species, which required an expanded repertoire of chemical and physical methods, could chemists envision a program of organic synthesis – to recompose organic substances directly from simple bodies via chemical forces, as Marcelin Berthelot characterized it in La chimie organique fondée sur la synthèse (1860). While Berthelot’s intense rivalry with Adolph Wurtz may be regarded as the fourth theoretical moment that preceded the articulation of structural chemistry, or classical organic chemistry, chemists’ central concern by this time lay no longer in naturalizing their laboratory products (Russell 1987, Rocke 2001, Kim 2005). The focus on these theoretical moments should make it clear that chemical theories in history were not limited to corpuscular ontologies.[6] A successful theoretical framework for chemistry such as the affinity table had to coordinate existing analytic methods to provide a coherent classification of their products and afford a reliable prediction of their behavior (Duncan 1962, Klein 1995, Kim 2003, pp. 111-159). Understanding this historically variable relationship between chemical methods, theories, and objects should sharpen our reflexivity on scientific practice to define the investigative frontier of historical epistemology, or "the discussion of matters of contemporary concern using the resources of history" (Tiles & Tiles 1993, p. 5). Reflections on the material contingency of chemical worlds should also help us to articulate historical ontology, or a critical reflection on how we identify and shape the objects of scientific/scholarly inquiry (Hacking 2002). These philosophical concerns in the critical mode differ significantly from the efforts at historicizing/socializing epistemology and ontology, which often require detailed historical/social reconstructions and are aptly named epistemography and ontography (Dear 2001, Lynch 2013).[7] 2. The Analytic-Synthetic Ideal: Methods of World-making and KnowingMaking a coherent chemical world out of heterogeneous nature, like other efforts at world-making and remaking, depended on the methods of decomposition and composition. Unlike painters or writers, however, chemists had to work collaboratively and communicate with each other at every step of their world-making and have left us a much more detailed record of their labors and mirages (Kim 1995). Chemists’ evolving laboratory methods and symbolic representations should thus offer philosophers an exceptionally rich resource to address Goodman’s important question, "how is world-making related to knowing?" (Goodman 1978, p. 1) Ethnographic sensitivity to chemists’ laboratory methods and products can become a powerful tool in probing the philosophical questions on plurality of worlds and their respective realness. It can also help us shape a historical epistemology that probes the interplay between "ways of knowing and ways of working", as John V. Pickstone (2011) put it. He aptly characterized their cumulative ‘nesting’ as a process of forming compounds out of elementary ideal types. Chemists’ own process of articulating and implementing the analytic-synthetic ideal of chemical substances, or knowing their composition, involved stabilizing various analytic methods, sorting out their differential products, and making them compatible with each other to read them as ‘natural kinds’ (Hendry 2012). In other words, the primary function of chemical theory consisted in coordinating diverse analytic methods to hybridize/naturalize their products. The analytic ideal of chemical elements was stabilized by seventeenth-century French chemists through their effort to correlate distillation products and philosophical reality for a public discourse of chemistry (Metzger 1969, Hannaway 1975, Kim 2001).[8] Distillation had become the representative method of vegetable analysis in pharmaceutical practice, at least in France. According to Étienne de Clave (1641, p. 260), fire resolved mixts into "five elements or first principles which were actually included and hidden in them: spirit or mercury, water or phlegm, sulphur or oil, salt, and earth". Chemists adapted philosophical systems to their laboratory practice, that is, to produce a ‘strangely modern’ definition of the element as "a simple body that actually enters into the mixture of compound bodies and to which they can be finally resolved" (ibid.). De Clave’s ‘true’ principles selectively combined Aristotelian four elements and Paracelsian tria prima to designate simple bodies that formed diverse mixts in various proportions (Kahn 2001). Despite the insurmountable difficulty in matching distillation products to the supposedly natural (philosophical) elements, this hybrid construction of the analytic/philosophical ideal helped French Paracelsians (chemical physicians) to create a mediating social and cognitive space of chemical theory between vulgar apothecaries and bookish physicians for a positive identity of chemistry and chemists (Kim 2003, pp. 17-63). The infusion of solution methods into pharmaceutical practice generated new demands for chemical theory, however. Originally deployed in metallurgical and alchemical practices, strong acids and alkalis promised a less destructive method than distillation to preserve the original components of bodies, but produced a different set of chemical components. In order to bridge the gap between solution and distillation analysis, Samuel Cottereau Du Clos at the Paris Academy of Sciences sought to moderate Robert Boyle’s corpuscular interpretation of chemical principles (Clericuzio 1990, 1993), to compete with Denis Dodart’s grand project on natural history (Stroup 1990, pp. 70-116), and to work out a philosophical vision (Kim 2003, pp. 48-52; Franckowiak 2011, Boantza 2013). In contrast, Claude Bourdelin continued with a systematic distillation of all known plants to discern their composition and medical virtue in concert with Dodart’s project. Such contested juxtaposition of distillation and solution methods, both of which produced morphological (rather than compositional) classifications of their products, made it extremely difficult to develop a coherent systematics of chemical bodies. A ‘salt’ was obtained in distillation method by dissolving the earthly residue and evaporating the solution. In solution analysis, salts referred to all acids, alkalis, and their combinations. Different laboratory procedures yielded different knowledge of chemical composition, which interfered with constructing a coherent laboratory reality and chemical theory. This was the juncture at which Wilhelm Homberg joined the Academy and sought to overcome the impasse with new instruments (Kim 2000, Principe 2001). In order to understand his instrumental intervention in chemical practice as well as his theoretical aspirations for chemical discourse, we must recognize his rather unconventional path to chemistry. Versed in the Boylean fashion of experimental philosophy, he turned to a powerful furnace – the Tschirnhaus burning glass – to forge a consistent identification and classification of distillation and solution products. He hoped that the burning glass would dissolve chemical bodies to their smallest corpuscles to provide a shared instrumental/philosophical foundation for an integrated theory and comprehensive classification of chemical bodies. He thus characterized pure fire as a dual agent – a tangible, corpuscular substance that composed other bodies and a universal solvent that dissolved all bodies to their ultimate constituents, or the long elusive alkahest. In a series of articles, titled ‘Essais de chimie’ (1702-9), Homberg sought to coordinate his systematic investigation for a consistent identification and comprehensive classification of chemical bodies and a new set of textbooks. Drastically simplified, his scheme would look like this (Table 1: if one supposed that the ‘true sulphur principle’ or the ‘matter of light’ could form a variety of sulphur principles (metallic, bituminous, vegetable) by combining with other bodies, these sulphur principles (the limits of actual chemical analysis) could in turn produce common mixts like metals, common sulphur, and vegetables. That is, one could not only provide a theoretically consistent identification and classification of chemical species, but also transmute them one into another.
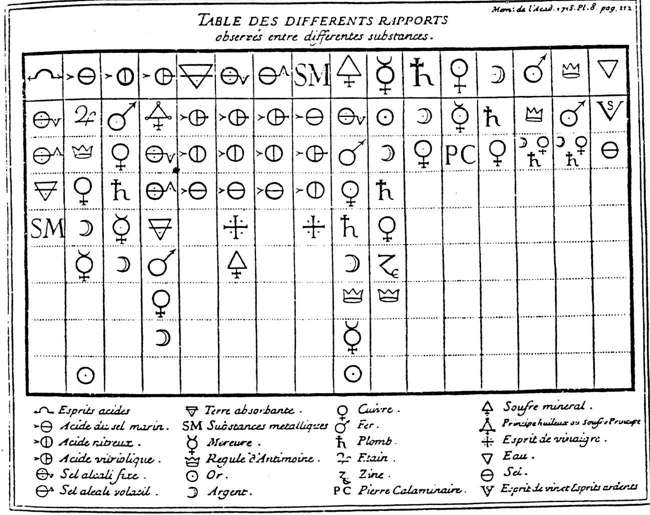
Homberg failed to produce his planned textbooks for a fundamental reform in chemical theory or practice, but his effort – instrumental and speculative/transmutationist – helped produce in the subsequent decades an algebraic ideal of analysis and synthesis for characterizing chemical substances. With the burning glass experiment, Homberg theorized that gold consisted of mercury, a metallic sulphur, and an earth. Common sulphur, analyzed with open flame, yielded an acid, an inflammable oil, and an earth. Étienne-François Geoffroy, who joined the Academy as Homberg’s student with more extensive background in pharmacy, not only recomposed common sulphur from its three decomposition products, but also tried to determine the composition of iron as consisting of the sulphur principle or an inflammable part, a vitriolic salt, and an earth. If he could elucidate the composition of all metals as sharing their sulphur principle with common sulphur, metals could be transformed into common sulphur. Such transformation would prove that the sulphur principle was the sole active principle in the universe. Geoffroy made the analytic/synthetic ideal of chemical principles/mixts explicit: One is never sure, in fact, of having decomposed a Mixt into its true principles until one can recompose it from the same principles. This reestablishment is not always possible. When it is not possible, it does not necessarily work against the analysis of the mixt, but when it is successful, the analysis is demonstrated. [Geoffroy 1704, p. 37] 3. Affinity Table: Representing a Laboratory WorldAlthough Homberg employed a hypothetical principle that required the rare burning glass, Geoffroy as an experienced apothecary tackled the transmutationist speculation with concrete chemical operations. After Homberg’s death, Geoffroy presented his Table des rapports (1718) (see Figure 1) to the Academy as a theoretical device that would help chemists to ‘discover’ and to ‘predict’ what happens when they mix several bodies in the solution of water or fire (Duncan 1962, Klein 1995). In visualizing chemical composition as the interplay of affinities, Geoffroy’s table indicates that solution methods became the representative analytic method by the early eighteenth-century to stabilize the objects of chemical inquiry, mostly salts (Holmes 1989a, Kim 2003, pp. 111-159). Instead of the five principles projected from the distillation method and naturalized by Aristotelian and Paracelsian philosophies, chemists could deal with the stable laboratory substances that combined or separated to illustrate the analytic-synthetic ideal of chemical species.[9] Their stability, including that of the sulphur principle (later identified as phlogiston – see the ‘Soufre principe’ in Figure 1), depended on chemists’ analytic capacity to isolate and characterize acids, alkalis and middle-salts across the boundaries of diverse analytic methods (Kim 2008). This was what a theory meant to Geoffroy – to offer a reasonable explanation of chemical operations across analytic boundaries – a working definition much more congenial to the epistemological culture of material sciences such as chemistry, medicine, and biology. Both the interpretation of experimental data and the design of new chemical experiments depended on the theoretical analysis of possible affinities.
Figure 1: Geoffroy’s affinity table (from Geoffroy 1718). Geoffroy’s affinity table offered a map of the chemical world constituted by operationally defined substances. It was a theoretical edifice that resulted from Homberg’s effort to ground chemical systematics on a set of coordinated analytic methods. Because the affinity table did not rely on any philosophical system or philosophical reality, it could organize chemical practice into two interlocking theory domains – composition and affinity. Its efficacy in guiding chemists’ laboratory practice can be easily discerned from its unruly expansion, as evidenced in Torbern Bergman’s table in the early 1780s, which prompted serious efforts to quantify affinities by Guyton de Morveau and others (Kim 2003, pp. 219-277). While the collaborative effort on chemical composition around Homberg provided a significant impetus to streamline chemical theory as it related to laboratory practice, what resulted was not a mindless implementation of his corpuscular speculation but a methodical intervention that stabilized an experimental program and theoretical representation of salts. Eighteenth-century French chemistry inherited a complex tradition that comprised corpuscular imagination and a stable repertoire of analytic methods, which they combined to produce a comprehensive theoretical structure that defined salts as stable objects of inquiry. The shift from the principalist to the affinity approach to composition took place in large part due to the change in chemists’ analytic methods that constituted their laboratory realities.[10] How early modern chemists naturalized their laboratory reality first by matching their analytic products to philosophical reality (five principles) and then by offering a map of their artificial reality (affinity table) is germane to our current discussions of instrumental reality (Ihde 1991, Radder 2003) and historical ontology. 4. Gases and the Redefinition of Chemical SpeciesThe second theoretical moment developed with Antoine Laurent Lavoisier’s systematic effort to incorporate the instruments of pneumatic and heat studies into chemical practice, which incorporated gases into chemical domain. Although historians and philosophers have studied Lavoisier’s extraordinary journey in detail (Guerlac 1961, Holmes 1998, Musgrave 1976, Thagard 1990, Pyle 2000), they do not yet appreciate how difficult it was to create the genus of gases and why it necessitated the elimination of phlogiston and a redefinition of chemical species. If the different kinds of air were simply added as individual chemical species in the way phlogistic chemists did with inflammable air, chemical reality would have comprised more species existing in the fixed physical states of solid, liquid, or gas. In contrast, Lavoisier defined a chemically distinct species as one that would not lose its properties (e.g. affinity) throughout what we now term as phase transitions, which produced a more coherent chemical reality that incorporated gases. He made a well-characterized chemical substance a permanent chemical species of which many individual bodies existed or could exist in three physical states. All these facts, which could be easily multiplied if necessary, give me full right to assume, as a general principle, that almost every body in nature is susceptible of three states of existence, solid, liquid, and aëriform, and that these three states of existence depend upon the quantity of caloric combined with the body. Henceforwards I shall express these elastic aëriform fluids by the generic term gas; and in each species of gas I shall distinguish between the caloric, which in some measure serves the purpose of a solvent, and the substance, which in combination with the caloric, forms the base of the gas. [Lavoisier 1965, p. 15, added emphasis] Lavoisier’s attack on phlogiston must be understood in this vein. As a matter of ontology or metaphysics, phlogiston and caloric shared many traits – both were subtle fluids endowed with irrepressible volatility or elasticity. As a matter of laboratory practice, however, phlogiston existed in the web of acid-alkali reactions that did not allow for a separate measurement and conceptualization of heat transactions (Kim 2008, 2011, Lewowicz 2011). British chemists’ identification of phlogiston with inflammable air may have been operationally easier for most chemists than Lavoisier’s metric alternative (Holmes 2000, Chang 2012, pp. 1-70), but it did not account for the heat transaction as a domain of instrumental control. Caloric was designed to stay outside the conventional chemical web to facilitate the metric measurement and algebraic representation of chemical actions. Lavoisier had an instrumental and classificatory rationale rather than metaphysical necessity to eliminate phlogiston and to advocate an algebraic nomenclature that made all gases into compounds of caloric and (often unknown) bases. His path to the chemical nomenclature reform was guided by metric (barometer, aerometer, thermometer, gasometer, and calorimeter) measurements, which supported his algebraic representation of chemical actions (Kim 2003, pp. 280-389). We can get a glimpse of Lavoisier’s laboratory reality that privileged chemical analysis from his carefully differentiated use of the words – ‘body’, ‘substance’, and ‘matter’ (Kim 1992a, pp. 71-77). In the Traité élémentaire de chimie (1789), ‘substance’ often designated a product of chemical analysis and served as a stable unit in his ‘methodical’ classification. Charcoal, when identified by its property of decomposing oxygen gas, had to be considered as a ‘simple combustible substance’ due to chemists’ inability to analyze it further, although he routinely used the phrase ‘combustible body’ on other occasions.[11] His table of ‘simple substances’ (other than the first five that could be regarded as the elements of all bodies – light, caloric, oxygene, azote, and hydrogene) mostly consisted of the salts that could no further be decomposed – those that could combine with the oxygen base [oxygène] to form acids (metallic and non-metallic) and those that could produce neutral salts by combining with acids. The acids, for example, are composed of two substances, of the order of those which we consider as simple; the one constitutes acidity, and is common to all acids, and from this substance, the name of the class or the genus ought to be taken: the other is peculiar to each acid, and distinguished it from the rest, and from this substance is to be taken the name of the species. [Lavoisier 1789, p. xxi; Lavoisier 1965, p. xxvii] Although Lavoisier’s oxygen theory of acids soon proved to be a hasty generalization, it still provided a simple principalist method of classifying acids based on their chemical composition. This point of simplicity in chemical classification was often lost to sophisticated chemists such as Richard Kirwan who tried to identify chemical species through their multi-faceted actions or affinities. If composition is indeed a neglected aspect of the Chemical Revolution, we must recognize at least two divergent approaches to composition – principalist vs. affinity (Siegfried & Dobbs 1968, Kim 2003, Chang 2011). The physical instruments Lavoisier designed and sought to implement in chemical practice refined the boundaries of chemical species, which maintained their identities throughout physical changes, to define pure substances through gravimetric and calorimetric composition. In so doing, he constructed a grid for classifying gases and vapors yet to be discovered. In other words, Lavoisier brought the chemistry of salts to its analytic conclusion by adding an instrumental measure of heat transactions to that of weight changes. As cumbersome and useless as it was, the calorimeter held out the promise that heat changes can be measured as easily as weight changes to completely quantify chemical actions. Lavoisier’s self-conscious ‘revolution in chemistry and physics’ did not radically transform the theoretical structure of salt chemistry. Instead, it utilized new physical instruments to redefine chemical species and thereby strengthened the analytic-synthetic ideal of chemical species. He invented a new chemical world mapped by the pure substances that could subsist throughout the phase transitions. 5. The Stoichiometric Atom and Constitutional TheoriesReinterpreting Lavoisier’s reform in terms of chemical species and their classification, or the physical methods that allowed their reification, would allow us to recognize the truly revolutionary contribution pneumatic studies made to the development of nineteenth-century chemistry – a new standard of analytic purity for acids and alkalis, which produced a sharper stoichiometric resolution of chemical species and operations. Historians and philosophers have largely missed this link because it was proposed by Richard Kirwan, a famous ‘loser’ in the conventional story of the Chemical Revolution.[12] His effort to quantify affinities focused on measuring the saturation capacities of acids and alkalis, an analytic frontier broadly shared by many contemporary chemists. In trying to measure them precisely, Kirwan enlisted marine acid air as the analytic standard (Kim 2003, pp. 268-277). Because gases could be obtained relatively free of impurities, full implementation of his proposal would have systematically enhanced the analytic purity of all chemical substances. This attention to exact quantitative relations of elements and compounds, which soon erupted as the Berthollet-Proust debate (Fujii 1986), paved the way for stoichiometry – not exactly the mathematical science of chemistry, as Jeremias Benjamin Richter (1792-3) intended in coining the term, but a systematic determination of the weight relations of chemical substances. The focus on stoichiometry can help us unpack the complex interaction between corpuscular speculations, analytic methods, and chemical theories during the early nineteenth century, which stabilized pure chemical species (Kim 1992a/b). While historians and philosophers of science have mostly subsumed chemical activity of this period under the banner of chemical atomism, largely to ensure Dalton’s relevance to contemporary chemistry as well as his place in history, the label works against their painstaking research and conclusion that chemists’ practices and conventions were not reducible to Dalton’s speculative ontology. Chemists consistently differentiated the law of constant and multiple proportions from the hypothesis of ontological atoms. Alan Rocke thus defines chemical atomism as a commitment to the existence of "a unique ‘atomic weight’, a chemically indivisible unit [of an element] that enters into combination with similar units of other elements in small integral multiples", to avoid "all reference to the ultimate structure of matter" (Rocke 1984, pp. 12-13). While this is an accurate assessment as relating to the combining units of chemical elements, it distorts nineteenth-century usage in which the ‘atom’ designated the definite combining weight of an element or a compound, or the ‘stoichiometric atom’ (Kim 1992b, pp. 397-409), which provided the quantitative measure of a distinct chemical species. Gay-Lussac’s ‘law’ of combining volumes provided an independent measure for the stoichiometric atom. It is very important to observe that in considering weights there is no simple and finite relation between the elements of any one compound; it is only when there is a second compound between the same elements that the new proportion of the element that has been added is a multiple of the first quantity. Gases, on the contrary, in whatever proportions they may combine, always give rise to compounds whose elements are multiple of each other by volume. [Gay-Lussac 1809, p. 15] In consolidating stoichiometry as an experimental frontier, Jacob Berzelius thus equated Dalton’s ‘atom’ with Gay-Lussac’s ‘volume’ to ground chemical investigation on the "well-constituted fact" of volume ratios rather than on Dalton’s mere "supposition" of atoms and arbitrary ‘rule of simplicity’ (Berzelius 1813, p. 445). He offered a pragmatic table of combining weights based on exact analysis (Melhado 1981, Melhado & Frängsmyr 1992). The importance of stabilizing the stoichiometric atom can be discerned from the multiplication of the physical methods developed to this end, although it remained difficult to correlate them for a universal measure, which fueled the ‘confusion’ over the constitution of organic bodies. For the purpose of this paper, it suffices to establish that chemists in the early nineteenth century endeavored to fix chemical species in a quantitative manner – elements or compounds – through the stoichiometric atom. This was a necessary step before they could attempt a classification of organic compounds. In contrast to inorganic compounds that consist of numerous elements in a small number of distinct proportions, organic compounds contain only a few elements that combine in an infinite variation of quantitative ratios. A refined analytic resolution was a necessary condition, therefore, for the identification and classification of organic species. In order to understand the development of organic chemistry in early nineteenth-century, we must accept classification as a serious theoretical edifice that reflects the contemporary epistemological culture of chemistry (Kim 1992a/b, Klein & Lefèvre 2007). Jean-Baptiste Dumas, whose migration from the atomic theory toward a ‘natural classification’ of organic substances opened the third theoretical moment, can guide us through the complex theoretical debate on organic constitution (Kim 1996, Bensaude-Vincent 1999). In his usage, the word ‘substance’ denoted a chemical species isolated through stoichiometric relations, losing its prior connotation as a common principle of bodies. A chemical substance thus would comprise individual ‘bodies’ that contained the chemical relationships elucidated through constitutional theories. His ‘natural classification’ would group organic bodies into species, genera, and families in terms of their dominant characteristics to predict their actions by chemical analogies (Dumas 1826). In trying to consolidate the program during the 1830s, however, he encountered numerous obstacles. Despite his need to characterize the ‘chemical atom’ based on chemical analogies without reference to the ontological atom, he had to find a way of fixing the stoichiometric atom consistently in order to discern distinct chemical species. Physical methods then available – vapor density, specific heat, and isomorphism – did not produce consistent numbers, which made it difficult to fix the stoichiometric atom for organic species. Multiple methods of determining the ‘atom’ left Dumas with multiple ontologies.[13] Here is therefore, it seems to me, the most probably consequences at which one arrives relative to the present state of our knowledge, in trying to account for the intimate constitution of bodies. Matter is formed of atoms. Specific heats teach us the relative weights of atoms of various kinds. Chemistry operates on the groups of the atoms of matter. These are the groups that produce combinations by uniting in different ratios according to the law of multiple proportions; these are those whose mutual displacements give reason for observing the rule of equivalents in reactions. Finally, the conversion into gas or vapor creates yet another molecular groups on which depend the laws observed by M. Gay-Lussac. [Dumas 1837, p. 282] Dumas’ laboratory experience and social acumen turned him toward the discourse of natural history and classification. His theory of substitution, or the type theory of organic constitution, was meant to provide "a natural classification of chemical substances that assigned proper places to them while predicting empty places to fill" – in the way the affinity table had worked for eighteenth-century chemists (Kim 2005, p. 175). Intense debate over the ‘constitution’ of organic bodies in the 1830s indicates a new character of chemical theory, however. The theory domains of composition and affinity established in the previous century by studying mineral salts no longer sufficed to differentiate and classify organic substances. Chemical practice relied now heavily on physical measurements just to determine the basic units of operation. Gone was the metaphysical discourse of philosophy that could supply the legitimating vocabulary of nature. If the moment reflected, as before, a master Academician’s conscious effort to consolidate his leadership by offering a comprehensive theoretical structure, Dumas could do so in the 1830s without relying on a philosophical paradigm. Chemistry had become a secure discipline with an established record of service to the state and a public claim for its scientific status, while philosophy faced an acute academic crisis (Kim 2009). Although Dumas chose for his inaugural lectures at the Collège de France the topic of ‘chemical philosophy’, perhaps to address the larger public that clamored for a unified representation of nature (Appel 1987), he sought to establish the ‘general principles’ of chemistry, or "to give the most general explication of chemical phenomena, to establish the liaison that exists between the observed facts and the cause of these facts". Rather than the ‘sterile conceptions’ of molecular mechanics or atomic ontology, he claimed, what all chemists shared was the "method […] an unlimited confidence accorded to experiment and a complete submission to the power of facts" that guided their "art of experimenting". The method of classification would guide chemists securely in their progression from a "hypothesis" (or an idea bordering on the purely speculative) to a "theory" (an idea supported by a group of facts), then to a "law" (a theory that attained a stronger degree of certainty through its ability to coordinate a broader range of facts) (Dumas 1837, pp. 1-4). His famous condemnation of the word ‘atom’ as a hypothetical concept that reached beyond the realm of experiment did not mean, however, a triumph of Comtean positivism (Bensaude-Vincent 1999). Dumas’ conscious effort to refashion chemical philosophy indicates instead chemistry’s confident stride in the period, in addition to the lack of a dominant philosophical discourse that could serve the purpose of social legitimation. Theoretical moments in French chemistry may be characterized as rhetorical moments when newly ascending master chemists sought to assert their authority by providing comprehensive theoretical direction. Lavoisier’s ghost helped such performative conjunctures throughout the nineteenth century, which in part accounts for Marcelin Berthelot’s grand vision of organic synthesis (Kim 1996, 2006). Bethelot’s intense competition with Adolphe Wurtz for the symbolic leadership of French chemistry again produced acrimonious debates over the function of chemical theory (Rocke 2000). More of a literary synthesizer than a laboratory innovator, Berthelot penned La chimie organique fondée sur la synthèse (1860) after he assumed the new chair of organic chemistry at the École de Pharmacie. He claimed that Lavoisier had defined chemistry as "the science of analysis" that pursued successive decompositions to discern simple bodies, but that analysis constituted only a half of the chemical problem. Once chemists attained simple bodies, they had to recompose the bodies they had destroyed through decompositions. It was this power of synthetic formation that gave chemistry its true character, Berthelot insisted, by distinguishing it from other natural sciences through the highest degree of certitude. Chemistry at its destination had to be "the science of synthesis", although he fell short of promising a complete reconstruction of natural bodies from chemical elements (Berthelot 1860, p. xi). While analytic methods separated material beings into immediate principles and then to elementary bodies, synthetic methods could only reverse the second step of reproducing the ‘chemically defined immediate principles’ from the elements, or the process governed by molecular forces. The 1500-page book brought Berthelot public acclaim and a chair of organic chemistry at the Collège de France in 1863. His lectures there, published as Leçons sur les methods générales de synthèse en chimie organique (1864), again raised rhetorical stakes of direct organic synthesis via simple and general methods as the cutting-edge science. German universities began to build new, palatial laboratories for organic chemistry at a record pace. The ‘quiet revolution’ that produced the structural chemistry by the 1860s took place elsewhere in Germany (Rocke 1993), however, while French chemists quibbled over metaphysical truth of Daltonian atoms (Needham 2004, 2008, Zwier 2011). With the dismal outcome in the Franco-Prussian War, they would launch a serious debate over the decline of French science (Rocke 1993, Gilpin 1968, Paul 1972). 6. ConclusionPhilosophers may not wish to bother with petty political intrigues, career motivations, national research styles, and imperial wars that spurred on French chemists’ pursuit of the analytic-synthetic ideal over two centuries. Rhetorical stakes of these theoretical moments provide valuable points of entry, however, to their thought processes and material manipulations that otherwise remain hidden. An archeology of empirical knowledge and lost material worlds requires paying attention to the methods that produced ephemeral beings and theories (Shapin & Schaffer 1985, Kim 2014) Most valuable in our effort to construct their genealogy is not what they publicly fought over – theories of composition, affinity, and constitution – but what they took for granted as the established methods of analysis. A history of chemistry conducted with ethnographic sensitivity to chemists’ contingent materiality and symbolic representations can offer valuable insights on how experimental sciences depend critically on the methods of inquiry (historical epistemology) that shape our laboratory objects as ‘natural’ entities (historical ontology). Philosophy of chemistry should be historical because chemical reality is a historically constructed one. This does not mean, however, that historians and philosophers of chemistry should spend all of their time on minutely historicizing chemical objects in their successive stages of evolution. Not only is this an impossible task but also it lacks philosophical purpose. The efforts at understanding and/or reproducing past material worlds and texts must not be an idle antiquarian pursuit for authenticity or a commitment to full-blown epistemological constructivism (Wink 2006) but an exercise in material hermeneutics to appropriate past material knowledge for a critical evaluation of how we do science in the present.[14] Modern chemistry provides us with a unique history of labor in which every laborer produces his own laboratory reality and representations of ‘nature’, unlike other artisanal productions (Kim 1995, Smith 2004). Chemists’ well-documented struggle to produce a coherent laboratory reality and to hybridize/naturalize it can help us understand how material knowledge is acquired through disciplined labor more than abstract ideas. In other words, history of chemistry can provide a deep reservoir of sources in articulating a philosophy of labor/technology/ knowledge, which should guide our uncertain steps toward an engineered bio- and eco-system. Historical ontology is not about reconstructing past objects through historical records, but a reflexive exercise on our scholarly endeavor that requires constant awareness and scrutiny of our own epistemological cultures. In this vein, it is important not to model these theoretical moments in the Kuhnian fashion. In identifying the "most obvious examples" of scientific revolutions, Thomas S. Kuhn (1962, p. 6) discerned four "defining characteristics": (1) the community’s rejection of once-honored scientific theory in favor of another incompatible with it; (2) a consequent shift in the problems available for scrutiny and in the standards that determine what should count as a legitimate problem or solution; (3) "a transformation in scientific imagination" that change the world within which scientific work is done; and (4) the controversies that ensue. In other words, his model for the progress of science was built around his understanding of the recent revolution in theoretical physics.[15] Chemical theories in history did not (perhaps still do not) function in the way histories of theoretical physics and their revolutions dictate (Rocke 2013, Needham 2014). Their task is not to represent nature as it always has been, but to engender a coherent chemical reality that can be made relevant to the representation of nature and society. The world chemists sought to make and describe has not been the immutable ‘nature’ at least for some time. Twentieth-century biologists in catching up with chemistry’s laboratory game, and less apologetic about their engineering goals, have offered us valuable insights to the process of making laboratory realities. Their notion of the ‘experimental system’ refers to an ensemble of materials, instruments, and techniques that configure a stable object of inquiry. Hans-Jörg Rheinberger reifies it as the ‘epistemological units’ that shape and reshape the ‘epistemic things’ of biological investigation. Experimental systems are the "genuine working units of contemporary research in which the scientific objects and the technical conditions of their production are inextricably interconnected" (Rheinberger 1997, p. 2). They sustain a field of inquiry by generating scientific novelties (phenomena or material entities), signifiers, and concepts. They generate epistemic objects that possess ‘characteristic, irreducible vagueness’ which are often ‘differentially reproduced’ by individual experimental systems. In other words, no two laboratories would deal with exactly the same experimental system or epistemic object. The notion of experimental system allows us to portray the complexity and autonomy of experimental practice and the constant displacements that take place in it. Intrinsically, experimental systems are constantly mutating, dynamic units less prone to historical fetish or antiquarian interests. As such, the notion should work well for characterizing the practice of modern chemistry (Kim 2006). In short, philosophy of chemistry requires a new framework outside conventional philosophy of science/physics based on the deeply held metaphysical assumption of immutable nature. It would be tedious to rehash the ongoing debate over the varieties of realism and pluralism, which do not target a field of practice that molds its own objects of inquiry. In characterizing the theoretical moments in history of chemistry, I am more interested in the complexity and anxiety they present to historical and philosophical investigation than in the notion of progress, either by accumulation or by revolution (Hoyningen-Huene 2008). Chemical reality is an evolving eco-system populated by the substances stabilized through a contingent repertoire of laboratory procedures which, in turn, were shaped by the material, social, and political conditions of history. A new philosophy of chemistry can begin only when we have a different history of chemistry, one that charts the historical development of the analytic and synthetic methods that produced on the one hand stable objects (substances) and, on the other hand, changing chemical theories. In order to develop as a field that could offer general perspectives on experimental sciences, it has to recognize the historicity of chemical entities and the peculiar epistemological culture of chemistry. AcknowledgementsThis paper was originally prepared for the 4th International Symposium on Structure-Property Relationship in Solid State Materials (Bordeaux, 2012). The author wishes to thank Dr. Antoine Villesuzanne for the invitation to address a diverse chemical audience as well as Dr. Carin Berkowitz at the Chemical Heritage Foundation for organizing the session ‘What is the Object of History of Chemistry?’ for the 7th British-North American Joint Meeting of the BSHS, CSHPS and HSS (Philadelphia, 2012). She is indebted to Dr. Joachim Schummer and two anonymous referees for their helpful comments on an earlier version. Notes[1] This recent definition differs significantly from the originary conception of Stéphane-Armand Nicolas Leduc, the author of La Biologie synthétique (1912), a futuristic title that parodied Berthelot’s La Synthèse chimique (1876) (Bensaude-Vincent 2009). [2] This is reflected in F.A. Paneth’s (2003) eloquent, albeit historically skewed, presentation of the duality of chemical elements as ‘basic substance’ and ‘simple substance’ in 1931. [3] The best-known case is Justus Liebig’s apparatus for organic analysis (Holmes 1989b, Brock 1997, Rocke 2000, Usselman 2003). On can also make a similar argument about the introduction of NMR as a method of identifying organic compounds. [4] It is important to differentiate laboratory methods or techniques from methodology or philosophical method (Brooke 1987). [5] While Immanuel Kant played a critical role in forging scientific [wissenschaftliche] philosophy through epistemological concerns, he did not recognize chemistry’s central role in defining modern science (Friedman 1992, Carrier 2001, Van Brakel 2006, Kim 2009). [6] This undercuts the Klein-Newman debate. Whether early modern chemistry was artisanal or scholarly, the identification of persistent components would have required stable methods (Klein 2007, 2011, 2012, Newman 2009). [7] Klein & Lefèvre’s (2007) ‘historical ontology’ of chemical objects has a stronger affinity with these STS approaches than with Foucault-inspired philosophical inquiries. [8] For earlier possibilities, see Newman 2006. [9] Klein (1994) characterizes this as the concept of chemical compound. [10] These two approaches are nicely abstracted and generalized in Chang 2011 as ‘principalism’ vs. ‘compositionism’. [11] Lavoisier’s careful differentiation of the terms was often lost in translation. Compare in this case Lavoisier 1789, p. 120 and 1965, p. 112. [12] Lavoisier largely missed this developing frontier, probably because of his principalist approach to composition, which created a blind spot in his classification (Klein & Lefèvre 2007). [13] The stoichiometric atoms deduced from vapor densities and specific heats, neither of which could be applied to all chemical substances, did not agree with those calculated from chemical analogies (Kim 1992b). [14] A notable effort to this end is Chang’s (2004) notion of ‘complementary science’. [15] For a different model, see Galison 1997. ReferencesAppel, T. A.: 1987, The Cuvier-Geoffroy Debate: French Biology in the decades before Darwin, Oxford: Oxford University Press. Baird, D.; Scerri, E. & McIntyre, L. (eds.): 2006, Philosophy of Chemistry: Synthesis of a New Discipline, Dordrecht: Springer. Bensaude-Vincent, B.: 1999, ‘Atomism and Positivism: A Legend about French Chemistry’, Annals of Science, 56, 81-94. Bensaude-Vincent, B. & Newman, W.R. (ed.): 2007, The Artificial and the Natural: An Evolving Polarity, Cambridge, MA: MIT Press. Bensaude-Vincent, B. & Simon, J.: 2008, Chemistry, the Impure Science, London: Imperial College Press. Bensaude-Vincent, B.: 2009, ‘Biomimetic Chemistry and Synthetic Biology: A Two-way Traffic across the Borders’, Hyle: International Journal for Philosophy of Chemistry, 15, 31-46. Berthelot, M.: 1860, Chimie organic fondée sur la Synthèse, Paris: Mallet-Bachelier. Berzelius, J.: 1813, ‘Essay on the cause of chemical proportions, and on some circumstances relating to them: together with a short and easy method of expressing them’, Annals of Philosophy, 22, 443-54. Boantza, V.D.: 2013, Matter and Method in the Long Chemical Revolution, Farnham: Ashgate. Brock, W.H.: 1997, Justus von Liebig: The Chemical Gatekeeper, Cambridge: Cambridge UP. Brooke, J.H.: 1987, ‘Methods and Methodology in the Development of Organic Chemistry’, Ambix, 34, 147-55. Carrier, M.: 2001, ‘Kant’s theory of matter and his views on chemistry’, in E. Watkins (ed.), Kant and the Sciences, Oxford: Oxford University Press, pp. 205-230. Chang, H.: 2004, Inventing Temperature: Measurement and Scientific Progress, New York: Oxford University Press. Chang, H.: 2011, ‘Compositionism as a Dominant Way of Knowing’, History of Science, 49, 247-68. Chang, H.: 2012, Is Water H2O? Evidence, Realism and Pluralism, Dordrecht: Springer. Clericuzio, A.: 1990, ‘A Redefinition of Boyle’s Chemistry and Corpuscular Philosophy’, Annals of Science, 47, 561-89. Clericuzio, A.: 1993, ‘From Van Helmont to Boyle’, British Journal for the History of Science, 26, 303-34. Clow, A. & Clow, N.L.: 1952, The Chemical Revolution: A Contribution to Social Technology, New York: Books for Libraries Press. Dear, P.: 2001, ‘Science Studies as Epistemography’, in Labinger, J.A. & H. Collins (ed.), The One Culture? A Conversation about Science, Chicago: University of Chicago Press, pp. 128-41. De Clave, E.: 1641, Nouvelle lumière philosophique des vrais principes et elemens de nature, & qualité d’iceux, Paris: O. de Varennes. Dumas, J.B.: 1826, ‘Mémoires sur quelques points de la théorie atomistique’, Annales de chimie et de physique, 33, 337-91. Dumas, J.B.: 1837, Leçons sur la philosophie chimique, Paris: Bechet jeune. Duncan, A.M.: 1962, ‘Some Theoretical Aspects of Eighteenth-Century Tables of Affinity’, Annals of Science, 18, 177-196, 217-32. Franckowiak, R.: 2011, ‘Mechanical and Chemical Explanations in Du Clos’ Chemistry’, Ambix, 58, 13-28. Friedman, M.: 1992, Kant and the Exact Sciences, Cambridge, MA: Harvard University Press. Fujii, K.: 1986, ‘The Berthollet-Proust Controversy and Dalton’s Chemical Atomic Theory, 1800-1820’, The British Journal for the History of Science, 19, 177-200. Gadamer, H.G.: 1993, Truth and Method, New York: Continuum. Galison, P.: 1997, Image and Logic: A Material Culture of Microphysics, Chicago: University of Chicago Press. Gardner, T.S. & Hawkins, K.: 2013, ‘Synthetic Biology: Evolution or Revolution? A Co-founder’s Perspective’, Current Opinion in Chemical Biology, 17, 871-7. Gay-Lussac, J.L.: 1809, ‘Mémoire sur la combinaison des substances gazeuses, les unes avec les autres’, Mémoires de la Société d’Arcueil, 2, 207-34. Geoffroy, E.F.: 1704, ‘Sur la récomposition du soufre’, Histoire de l’Académie royale des sciences, 37-9. Geoffroy, E.F.: 1718, ‘Des différents rapports observés en Chymie entre différentes substances’, Mémoires de l’Académie royal des sciences, 256-69. Gilpin, R.: 1968, France in the Age of the Scientific State, Princeton: Princeton University Press. Goodman, N.: 1978, Ways of World-making, Indianapolis: Hackett. Guerlac, H.: 1961, Lavoisier – The Crucial Year: The background and origin of his first experiments on combusion in 1772, Ithaca: Cornell University Press. Hacking, I.: 2002, Historical Ontology, Cambridge, MA: Harvard University Press. Hannaway, O.: 1975, The Chemists and the Word: The Didactic Origins of Chemistry, Baltimore: Johns Hopkins University Press. Harris, M.L.: 2008, ‘Chemical Reductionism Revisited: Lewis, Pauling and the Physico-Chemical Nature of the Chemical Bond’, Studies in History and Philosophy of Science, 39, 78-90. Hendry, R.F.: 2012, ‘Chemical Substances and the limits of pluralism’, Foundations of Chemistry, 4, 55-68. Holmes, F.L.: 1989a, Eighteenth-Century Chemistry as an Investigative Enterprise, Berkeley: University of California Press. Holmes, F.L: 1989b, ‘The Complementarity of Teaching and Research in Liebig’s Laboratory’, Osiris, 5, 121-64. Holmes, F.L.: 1996, ‘The Communal Context for Etienne-Francois Geoffroy’s Table des rapports’, Science in Context, 9, 289-311. Holmes, F.L.: 1998, Antoine Lavoisier, the Next Crucial Year: or the sources of his quantitative method in chemistry, Princeton: Princeton UP. Holmes, F.L.: 2000, ‘The ‘Revolution in Chemistry and Physics’: Overthrow of a Reigning paradigm or Competition between Contemporary Research Programs?’, Isis, 91, 735-53. Hoyningen-Huene, P.: 2008, ‘Thomas Kuhn and the chemical revolution’, Foundations of Chemistry, 10, 101-115. Ihde, D.: 1991, Instrumental Realism, Bloomington: Indiana University Press. Kahn, D.: 2001, ‘Entre atomisme, alchimie et théologie’, Annals of Science, 58, 241-286. Kim, M.G.: 1992a, ‘The Layers of Chemical Language I: Constitution of Bodies vs. Structure of Matter’, History of Science, 30, 69-96. Kim, M.G.: 1992b, ‘The Layers of Chemical Language II: Stabilizing Atoms and Molecules in the Practice of Organic Chemistry’, History of Science, 30, 397-437. Kim, M.G.: 1995, ‘Labor and Mirage: Writing the History of Chemistry’, Studies in History and Philosophy of Science, 26, 155-65. Kim, M.G.: 1996, ‘Constructing Symbolic Spaces: Chemical Molecules in the Académie des Sciences’, Ambix, 43, 1-31. Kim, M.G.: 2000, ‘Chemical Analysis and the Domains of Reality: Wilhelm Homberg’s Essais de Chimie, 1702-1709’, Studies in History and Philosophy of Science, 31, 37-69. Kim, M.G.: 2001, ‘The Analytic Ideal of Chemical Elements: Robert Boyle and the French Didactic Tradition of Chemistry’, Science in Context, 13, 361-95. Kim, M.G.: 2003, Affinity, That Elusive Dream: A Genealogy of the Chemical Revolution, Cambridge, MA: MIT Press. Kim, M.G.: 2005, ‘Lavoisier, the Father of Chemistry?’, in: M. Beretta (ed.) Lavoisier in Perspective, Munich: Deutsches Museum, pp. 167-91. Kim, M.G.: 2006 ‘Experimental Systems and Theory Domains in Pre-Lavoisian Chemistry’, in: I. Malaquias, E. Homburg & M.E. Callapez (eds.), Chemistry, Technology and Society: Proceedings of 5th International Conference on History of Chemistry, Aveiro: Sociedade Portuguesa de Química, pp. 468-82. Kim, M.G.: 2008, ‘The ‘Instrumental’ Reality of Phlogiston’, Hyle: International Journal for Philosophy of Chemistry, 14, 27-51. Kim, M.G.: 2009, ‘A Historical Atlas of Objectivity’, Modern Intellectual History, 6, 569-96. Kim, M.G.: 2011, ‘From Phlogiston to Caloric: Chemical Ontologies’, Foundations of Chemistry, 13, 201-22. Kim, M.G.: 2014, ‘Archeology, Genealogy, and Geography of Experimental Philosophy’, Social Studies of Science, 44, 150-62. Klein, U.: 1994, ‘Origin of the Concept of Chemical Compound’, Science in Context, 7, 163-204. Klein, U.: 1995, ‘E.F. Geoffroy’s Table of different ‘Rapports’ observed between different chemical substances – A Reinterpretation’, Ambix, 42, 79-100. Klein, U.: 2007, ‘Styles of Experimentation and Alchemical Matter Theory in the Scientific Revolution’, Metascience, 16, 247-256. Klein, U.: 2012, ‘Objects of inquiry in classical chemistry: material substances’, Foundations of Chemistry, 14, 7-23. Klein, U. & Lefèvre, W.: 2007, Materials in Eighteenth-Century Science: Historical Ontology, Cambridge, MA: MIT Press. Kuhn, T.S.: 1952, ‘Robert Boyle and Structural Chemistry in the Seventeenth Century’, Isis, 43, 12–36. Kuhn, T.S.: 1962, The Structure of Scientific Revolutions, Chicago: University of Chicago Press Lavoisier, A.L.: 1789, Traité élémentaire de chimie, Paris: Cuchet. Lavoisier, A.L.: 1965, Elements of Chemistry, New York: Dover Press. Lewowicz, L.: 2011, ‘Phlogiston, Lavoisier and the purloined referent’, Studies in History and Philosophy of Science, 42, 436-44. Lynch, M.: 2013, ‘Ontography: Investigating the production of things, deflating ontology’, Social Studies of Science, 43, 444-62. Melhado, E.M. & Frängsmyr, T. (eds.): 1992, Enlightenment Science in the Romantic Era: The Chemistry of Berzelius and Its Cultural Setting, Cambridge: Cambridge University Press. Melhado, E.M.: 1981, Jacob Berzelius, the emergence of his chemical system, Madison: University of Wisconsin Press. Metzger, Hélène.: 1969, Les Doctrines chimiques en France du début du XVIIe à la fin du XVIIIe siècle, new edn., Paris: Albert Blanchard. Musgrave, A.: 1976, ‘Why Did Oxygen Supplant Phlogiston? Research Programmes in the Chemical Revolution’, in: C. Howson (ed.), Method and Appraisal in the Physical Sciences, Cambridge: Cambridge University Press, pp. 181-209. Needham, P.: 2004, ‘Has Daltonian Atomism provided Chemistry with Any Explanations?’, Philosophy of Science, 71, 1038-47. Needham, P.: 2008, ‘Resisting Chemical Atomism: Duhem’s Argument’, Philosophy of Science, 75, 921-31. Needham, P.: 2014, ‘Nineteenth-Century Chemical Atomism’, Foundations of Chemistry, 16, 165-7. Newman, W.R.: 1996, ‘The Alchemical Sources of Robert Boyle’s Corpuscular Philosophy’, Annals of Science, 53, 567-85. Newman, W.R.: 2006, Atoms and Alchemy: Chymistry and the Experimental Origins of the Scientific Revolution, Chicago: The University of Chicago Press. Newman, W.R.: 2009, ‘Alchemical Atoms or Artisanal ‘Building Blocks’?: A Response to Klein’, Perspectives on Science, 17, 212-31. Paneth, F.A.: 2003 [1931], ‘The Epistemological Status of the Chemical Concept of Element’, Foundations of Chemistry, 5, 113-45. Paul, H.W.: 1972, ‘The Issue of Decline in Nineteenth-Century French Science’, French Historical Studies, 7, 416-50. Pickstone, J.V.: 2011, ‘A Brief Introduction to Ways of Knowing and Ways of Working’, History of Science, 49, 234-45. Principe, L.M.: 2001, ‘Wilhelm Homberg, Chymical Corpuscularianism and Chrysopoeia in the Early Eighteenth Century’, in: C. Luthy et al. (eds.), Late Medieval and Early Modern Corpuscular Matter Theories, Leiden: Brill, pp. 535-56. Pyle, A.: 2000, ‘The Rationality of the Chemical Revolution’, in: R. Nola & H. Sankey (eds.), After Popper, Kuhn and Feyerabend, Dordrecht: Kluwer, pp. 99-124. Radder, H. (ed.): 2003 The Philosophy of Scientific Experimentation, Pittsburgh: University of Pittsburgh Press. Rheinberger, H-J.: 1997, Toward a History of Epistemic Things: Synthesizing Proteins in the Test Tube, Stanford: Stanford University Press. Richter, J.B.: 1792-3, Anfangsgründe der Stöchyometrie oder Messkunst chymischer Element, 3 vols., Silesia. Rocke, A.J.: 1984, Chemical Atomism in the Nineteenth Century: From Dalton to Cannizzaro, Columbus: Ohio State University Press. Rocke, A.J.: 1993, The Quiet Revolution: Hermann Kolbe and the Science of Organic Chemistry, Berkeley: University of California Press. Rocke, A.J.: 2000, ‘Organic Analysis in Comparative Perspective: Liebig, Dumas, and Berzelius, 1811-1840’, in: F. Holmes & T. Levere (eds.) , Instruments and Experimentation in the History of Chemistry, Cambridge, MA: MIT Press. Rocke, A.J.: 2001, Nationalizing Science: Adolphe Wurtz and the Battle for French Chemistry, Cambridge, MA: MIT Press. Rocke, A.J.: 2010, Image and Reality: Kekulé, Kopp and the Scientific Imagination, Chicago: University of Chicago Press. Rocke, A.: 2013, ‘What did ‘theory’ mean to nineteenth-century chemists?’, Foundations of Chemistry, 15, 145-156. Rorty, R.: 1979, Philosophy and the Mirror of Nature, Princeton: Princeton University Press. Russell, C.A.: 1987, ‘The Changing Role of Synthesis in Organic Chemistry’, Ambix, 34, 169-80. Schummer, J.: 2006, ‘The Philosophy of Chemistry: From Infancy Toward Maturity’, in: D. Baird et al. (eds.), Philosophy of Chemistry: Synthesis of a New Discipline, Springer, pp. 19-39. Shapin, S. & Schaffer, S.: 1985, Leviathan and the Air-Pump, Princeton: Princeton University Press. Sigfried, R. & B.J. Dobbs.: 1968, ‘Composition: A Neglected Aspect of the Chemical Revolution, Annals of Science, 24, 275-93. Simon, J.: 2005, Chemistry, Pharmacy and Revolution in France, 1777-1809, Farnham: Ashgate. Smith, P.: 2004, The Body of the Artisan: Art and Experience in the Scientific Revolution, Chicago: University of Chicago Press. Stroup, A.: 1990, A Company of Scientists: Botany, Patronage, and Community at the Seventeenth-Century Parisian Royal Academy of Sciences, Berkeley: University of California Press. Thagard, P: 1990, ‘Conceptual Structure of the Chemical Revolution’, Philosophy of Science, 57, 183-209. Tiles, M. & Tiles, J.: 1993, An Introduction to Historical Epistemology: The Authority of Knowledge, Cambridge: Blackwell. Usselman, M.C.: 2003, ‘Liebig’s Alkaloid Analyses: The Uncertain Route from Elemental Content to Molecular Formulae’, Ambix, 50, 71-89. Van Brakel, J.: 2006, ‘Kant’s Legacy for the Philosophy of Chemistry’, in: D. Baird et al. (eds.) Philosophy of Chemistry: Synthesis of a New Discipline, Dordrecht: Springer, pp. 69-91. Wink, D.: 2006, ‘Connections between pedagogical and epistemological constructivitism: questions for teaching and research in chemistry’, Foundations of Chemistry, 8, 111-51. Zwier, K.R.: 2011, ‘Dalton’s Chemical Atoms versus Duhem’s Chemical Equivalents’, Philosophy of Science, 78, 842-853. Mi Gyung Kim: |
||||||||||||||||